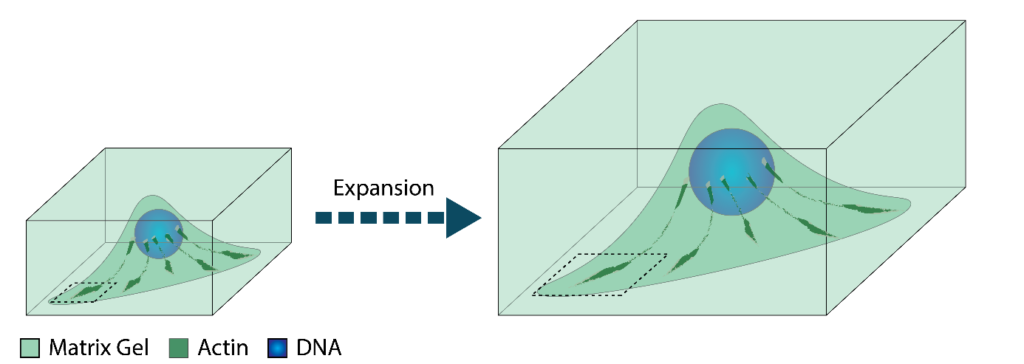
The resolution of optical microscopy, including fluorescence microscopy is limited to approximately 200 nm, due to the properties of light. However, new techniques have been developed that can circumvent these hard limits. These techniques are collectively known as super-resolution microscopy and require advanced and specific microscopes. An alternative method to visualise details in cells that are too small to resolve is to physically increase the size of sample, known as expansion microscopy (ExM). In this technique, samples are embedded in a gel, the cell content, including fluorescently labeled proteins or other biomolecules is then cross-linked to the gel and digested. Finally the gel is allowed to expand 4x to 10x. Microscopic imaging such an expanded sample then reveals details that initially were too small even when they are too small to visualise by super-resolution microscopy techniques. One potentially very promising application is to combine ExM with a so called pan-staining, which aims to label every protein in a cell with a fluorophore. In regular microscopy such staining will yield no discernable features since they lack resolution. However, using ExM several research groups have recently successfully applied pan-staining and claimed to achieve electron microscope (EM) resolution. This project involves setting up ExM and pan-staining in our lab to investigate focal adhesions. Focal adhesions are structures by which cells attach to a surfaces and move on it. We have already observed distinctive features of focal adhesions in EM and aim to observe the same using ExM and pan-staining. Additionally we want to combine this technique with regular staining of proteins to go beyond the capabilities of EM in which specific labeling of proteins is difficult. The aim of this project is to bridge the resolution gap between EM and light microscopy.
Contact Information:
Erasmus Optical Imaging Center
www.erasmusoic.nl
Johan Slotman (j.slotman [a] erasmusmc.nl)
Tel: 010-7037644

